A small, colourful fish from southern Africa has just upended one of science’s most basic assumptions about growing old, and the implications may reach all the way to the data on your smartwatch.
Researchers at Stanford University have published a study in the journal Science showing that the daily behaviour of individual animals can predict how long they will live, often well before midlife has passed. The finding emerged from what may be the most comprehensive surveillance project ever conducted on a living creature: 81 fish filmed around the clock, from birth to natural death, generating billions of video frames.
The animal at the centre of the experiment is the African turquoise killifish (Nothobranchius furzeri), a small, vividly coloured species native to the ephemeral water pools of Zimbabwe and Mozambique.

These pools exist for only a few months each year during the rainy season, which has driven the killifish to evolve one of the most compressed lifespans of any vertebrate, typically four to eight months in the laboratory. That short life is what makes it so useful to scientists studying aging. A researcher can watch an entire vertebrate lifespan unfold in less time than it takes most graduate students to finish a semester.
Watching every moment
The research team, led by postdoctoral scholars Claire Bedbrook and Ravi Nath, built an automated system in which each fish lived alone in a small tank under a ceiling-mounted camera running at 20 frames per second. Every feeding was handled by programmable dispensers. There was no off switch. From the moment each fish entered the tank at puberty, about three to four weeks of age, until the moment it died, every movement was recorded.
From that footage, the team extracted 100 distinct “behavioural syllables,” the basic repeating units of how a fish moves and rests. These included measures of posture, swimming speed, rest duration, and the timing of activity across the day-night cycle.
“Behaviour turns out to be an incredibly sensitive readout of aging,” Nath said in a statement from Stanford’s Wu Tsai Neurosciences Institute. “You can look at two animals of the same chronological age and see from their behaviour alone that they’re aging very differently.”
The sleep signal
The clearest early warning sign was sleep. Fish that went on to live shorter lives began sleeping not just at night but increasingly during the day as young adults, long before any outward signs of aging appeared. Fish that ultimately lived longer kept their activity concentrated in the daylight hours, sleeping mainly at night.
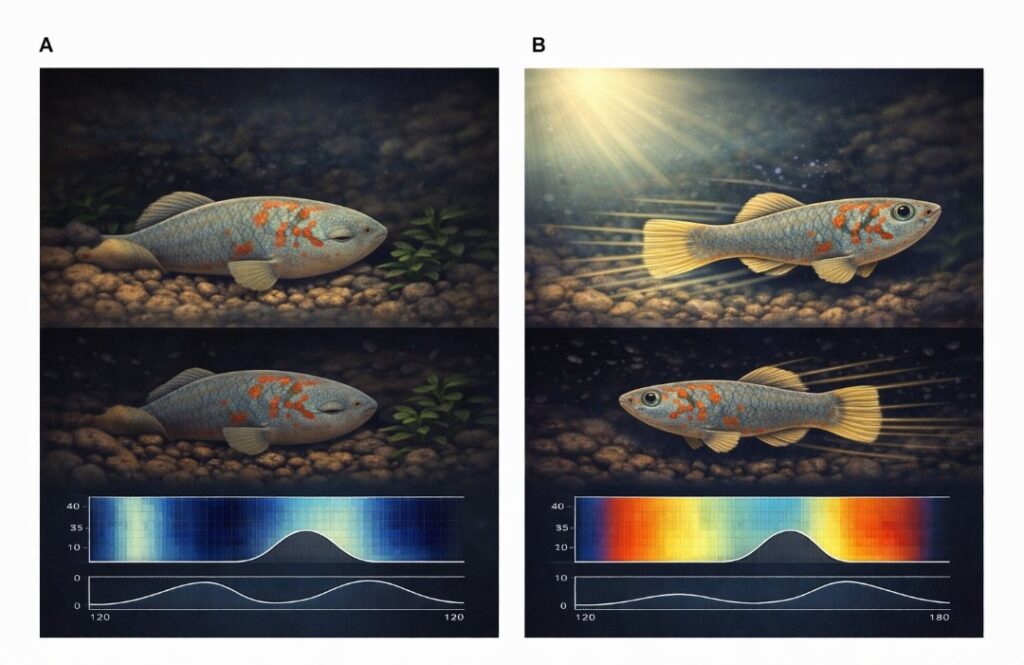
But sleep was not the only clue. Long-lived fish also swam with greater vigour. When they darted around their tanks, they reached higher peak speeds. They were more active overall during the day. Using machine-learning models trained on the video data, the researchers found that just a few days of behavioural data collected from a fish in early midlife, around 70 to 100 days of age, was enough to accurately forecast how long that fish would ultimately live.
“Behavioural changes pretty early on in life are telling us about future health and future lifespan,” Bedbrook said in the statement.
Aging does not happen gradually
Perhaps the most surprising finding was structural rather than predictive. The researchers expected aging to unfold as a slow, steady decline, each day slightly worse than the last. Instead, they found something quite different.
“Instead, animals stay stable for long periods and then transition very quickly into a new stage,” Bedbrook said. Each fish underwent between two and six rapid behavioural shifts across its adult life, each lasting only a few days, followed by longer stable periods that could stretch for weeks. Crucially, fish progressed through these stages in order rather than moving back and forth between them.
One way to visualise it: like a Jenga tower, the fish’s behavioural patterns stayed intact through many small changes, until a single transition forced a rapid reshuffling into a new and less stable state.
This “stepwise architecture” of aging, as the researchers describe it, mirrors recent findings in human biology suggesting that aging markers in people change in waves, particularly around midlife and older age, rather than continuously.
What the body was doing at the same time
The team did not stop at behaviour. At the point in each fish’s life when its behaviour became reliably predictive of lifespan, the researchers examined gene activity across eight different organs. Rather than looking for individual genes, they looked for coordinated shifts across groups of genes working on shared biological processes.
The clearest differences appeared in the liver. Genes involved in protein production and cellular maintenance were more active in fish on shorter aging paths. The fish that looked different on camera were running different biological programs inside their bodies at the same moment.
The team also tested whether aging paths could be altered. In a separate group of fish subjected to dietary restriction, fewer and earlier feedings each day, behavioural aging trajectories shifted, raising the possibility that intervention, not just prediction, may eventually be in reach.
What this means for humans
The researchers are measured about translating fish findings directly to people. Killifish and humans share fundamental biological features, including complex brains, but the two species diverged hundreds of millions of years ago. Still, the parallels are striking enough that the team raises the possibility openly.
“With the rise of wearables and long-term tracking in humans, I’m excited to see whether the same principles, early predictors, staged aging, divergent trajectories, hold true in people,” Bedbrook said.
In humans, sleep quality and the timing of the sleep-wake cycle are already known to deteriorate with age, and those changes have been linked to cognitive decline and neurodegenerative disease. Nath aims to explore whether sleep itself can be manipulated to slow aging in fish, and whether intervening before the first behavioural transition occurs can alter an individual’s trajectory.
Bedbrook explained to BBC Science Focus that since smartwatches are already tracking movement and sleep around the clock, scientists may soon be in a position to “understand and quantify where an individual is on their journey from adulthood to death.”
The study was conducted only in male killifish. Whether the same patterns hold in females remains to be tested.
Both Bedbrook and Nath are moving to Princeton University later this year to open their own laboratories, carrying the killifish system and its questions with them. Separately, Karl Deisseroth’s lab at Stanford is pursuing tools to monitor brain activity continuously alongside behaviour in the same animals, a step that could reveal whether the brain leads or follows the body as each aging transition unfolds.
Sources
The study was published in Science on March 12, 2026, under the title “Lifelong behavioral screen reveals an architecture of vertebrate aging.” It was supported by the Knight Initiative for Brain Resilience at Stanford’s Wu Tsai Neurosciences Institute.
Quotes in this article are drawn from press releases and statements issued by Stanford University and its Wu Tsai Neurosciences Institute, and from an interview published by BBC Science Focus, in March 2026.

Jane holds a BSc in Biology from the University of Regina and a Master of Science in Bioscience, Technology and Public Policy from the Univesity of Winnipeg. Her reporting interests include Life Sciences, Physical Sciences and the Cosmos.

