Parasitic wasps are nature’s most sinister puppeteers. They don’t just kill their hosts. They reprogram them.
Female wasps inject eggs into living caterpillars. The larvae grow inside, feeding on the host from within. But here’s the twist: before the host dies, the wasp sterilizes it first.
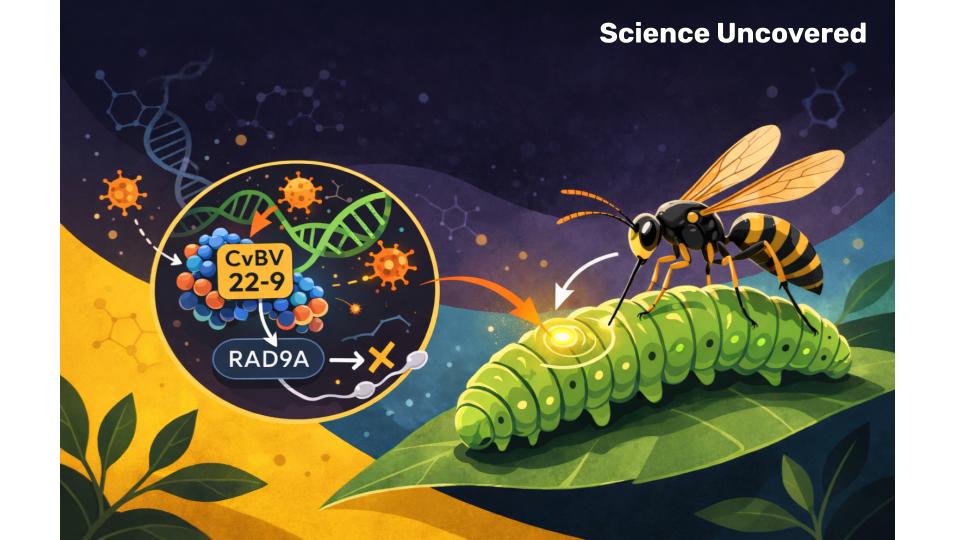
A new study [1] reveals exactly how they pull off this biological heist. The weapon? A domesticated virus carrying a molecular saboteur disguised as a helpful enzyme.
The Setup
Meet Cotesia vestalis and Microplitis manilae – two species of endoparasitoid wasps. They target caterpillars like diamondback moths and fall armyworms, both notorious agricultural pests.
When a female wasp finds a suitable host, she doesn’t just lay eggs. She delivers a biological payload: eggs plus virus particles. Not just any virus—a bracovirus that’s been integrated into the wasp’s own DNA for millions of years.
This virus is inherited. Passed down through generations like eye color or wing shape. It’s no longer an infection. It’s a weapon.

Tha parasitic wasp Trioxys complanatus is a biological control agent introduced to combat the spotted alfalfa aphid. Image credit: CSIRO
The Fake Enzyme
Inside the bracovirus genome lies a gene called CvBV_22-9. It codes for a protein that looks like a protein tyrosine phosphatase (PTP), an enzyme that normally removes phosphate groups from other proteins to regulate cell function.
But CvBV_22-9 is a fraud. A pseudophosphatase.
It has the shape. It has the binding sites. But it doesn’t do what phosphatases do. Instead, it does something far more devious.
The Target
The viral protein homes in on Rad9A, a crucial component of the cell’s quality control system. Rad9A is part of the 9-1-1 complex, which acts like a smoke detector for DNA damage.
In developing testes, cells divide rapidly to produce sperm. Rad9A keeps watch. It ensures everything runs smoothly. If DNA gets damaged, Rad9A hits the brakes.
When CvBV_22-9 binds to Rad9A, it doesn’t remove phosphate groups. It doesn’t “turn off” the protein in the normal sense.
It triggers a death signal.
The Kill Switch
The binding activates apoptosis – programmed cell death. Testicular cells begin to self-destruct en masse.
No testes. No sperm. No reproduction.
The caterpillar is chemically castrated.
Researchers at Zhejiang University, led by Zhizhi Wang, documented 50-70% reduced testis growth in parasitized larvae. The host stays alive—but its reproductive future is erased.
Proof in the Lab
The team used CRISPR gene editing to delete Rad9A from moth genomes. The results were dramatic.
Embryos died. Or if they survived, they had malformed testes. They couldn’t reproduce—exactly like parasitized caterpillars.
The researchers then tested the system in fruit flies (Drosophila melanogaster). When they forced fly testes to produce CvBV_22-9, cells started dying.
Reduce Rad9A levels at the same time? The destruction intensified.
The vulnerability isn’t unique to moths. It’s shared across insect groups. The wasps have found a universal weak point.
Why It Matters
This isn’t just a fascinating piece of natural history. It’s a window into molecular manipulation.
The diamondback moth and fall armyworm cause billions in crop damage worldwide. Understanding how parasitoid wasps naturally control these pests could revolutionize biocontrol.
Imagine engineered proteins that target pest reproduction without harming beneficial insects. Precision warfare at the cellular level.
The Bigger Picture
What makes this study remarkable is how it blurs the line between parasite and virus.
The bracovirus isn’t an infection anymore. It’s a tool. Domesticated over millions of years. The wasps carry viral genes in their own chromosomes and deploy them like guided missiles.
The virus particles can’t reproduce on their own—the wasp stripped out those genes long ago. All that remains is the weaponry.
Questions Remain
How exactly does the CvBV_22-9/Rad9A interaction trigger cell death without changing phosphorylation? The mechanism is still unclear.
Are there other pseudophosphatases lurking in parasitic systems? Probably. We’re just starting to look.
And here’s the really wild part: this entire system – virus domestication, molecular sabotage, reproductive manipulation, evolved naturally. No lab required.
Nature has been engineering biological weapons for millions of years. We’re only now learning to read the blueprints.
The Takeaway
Parasitoid wasps have turned viral infection into inheritance. They’ve converted a pathogen into a precision tool. And they’ve found a way to chemically castrate their hosts using a fake enzyme that exploits a universal cellular checkpoint.
It’s manipulation. It’s elegance. It’s evolution doing what it does best—finding the most ruthlessly efficient solution.
The caterpillars never stood a chance.
References
[1] Gao, H., Guo, M., Yang, X., et al. (2026). Parasitic castration by a viral protein tyrosine phosphatase targeting the host cell cycle checkpoint protein Rad9A. Proceedings of the National Academy of Sciences, 123(7), e2524949123.
[2] Nature Research Highlight (February 13, 2026). “Parasitic wasps use tamed virus to castrate caterpillars” Nature Biological Sciences.

Jane holds a BSc in Biology from the University of Regina and a Master of Science in Bioscience, Technology and Public Policy from the Univesity of Winnipeg. Her reporting interests include Life Sciences, Physical Sciences and the Cosmos.