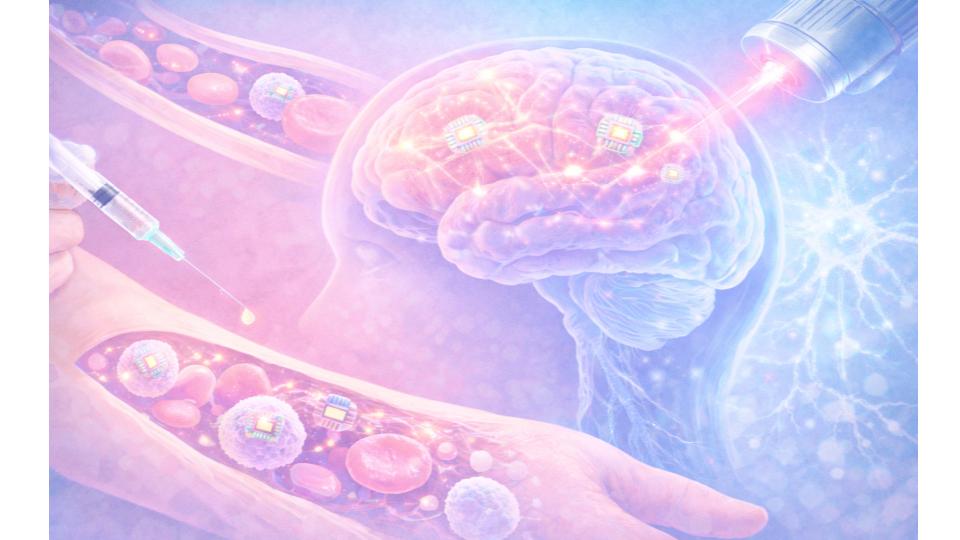
Microscopic electronic devices attach to immune cells, travel through the bloodstream, and self-implant in targeted brain regions without surgery, mouse experiments show.
CAMBRIDGE, Mass. — Researchers at the Massachusetts Institute of Technology have developed electronic implants small enough to travel through the bloodstream and embed themselves in brain tissue without surgery, according to a study published in November 2025 in Nature Biotechnology.
The devices, described by the research team as “circulatronics,” attach to immune cells called monocytes, which naturally cross the blood-brain barrier during inflammation. Once inside the brain, the implants receive power wirelessly via near-infrared light beamed through the skull and deliver targeted electrical stimulation to surrounding neurons.
In mouse experiments, the devices successfully self-implanted in a deep brain region involved in movement control, integrated among neurons, and caused no detectable cell death. The team has spun out a startup, Cahira Technologies, with the goal of reaching human trials within three years.
“This is a platform technology,” said Deblina Sarkar, who led the research at the MIT Media Lab. “It may be employed to treat multiple brain diseases and mental illnesses. And it is not just confined to the brain, it could also be extended to other parts of the body.”
The delivery problem the design solves
Conventional deep brain stimulation requires a neurosurgeon to drill into the skull, thread electrodes several centimeters into brain tissue, and implant a pulse generator under the collarbone. The procedure can cost more than $100,000 and carries significant surgical risk. As a result, it remains inaccessible to many patients worldwide.
The MIT approach uses the body’s own cellular machinery instead. The blood-brain barrier, a tightly sealed wall of cells surrounding brain blood vessels, blocks most foreign substances from entering brain tissue. It is a major reason many promising neurological drugs fail during clinical trials.
Monocytes, a type of immune cell, naturally cross the barrier when the brain becomes inflamed, as occurs in Alzheimer’s disease, multiple sclerosis, stroke, and brain tumors. Sarkar’s team attached their microscopic electronic devices to monocytes before injection.
“The living cells camouflage the electronics so they aren’t attacked by the immune system,” Sarkar said. “They can travel seamlessly through the bloodstream.”
Device design and function
Each implant is built from organic semiconducting polymers layered between thin metallic films. The devices are smaller than a human cell.
Despite their size, they function as complete wireless electronic systems capable of receiving power remotely and delivering focused electrical stimulation to nearby neurons.
Once monocytes carry the implants across the blood-brain barrier and into inflamed tissue, an external transmitter beams near-infrared light through the skull. The devices harvest the light as power. No implanted battery, no wires, and no surgical access are required.
Because the implants travel wherever immune cells migrate, they can distribute across an entire affected brain region rather than stimulating a single point as surgically implanted electrodes do.
Potential applications and current limitations
The research team identified glioblastoma, Alzheimer’s disease, Parkinson’s disease, and chronic pain as potential target applications. The approach is also being investigated for use outside the brain in other parts of the body where immune cell trafficking occurs.
All results so far come from mouse experiments. Several key questions remain unresolved, including long-term safety in humans, how precisely stimulation levels can be controlled, what happens to the implants after inflammation subsides, and the regulatory pathway required for clinical approval.
Many promising neurological therapies have failed during the transition from animal models to human patients.
Cahira Technologies, the startup founded by Sarkar’s team to commercialize the platform, is targeting human trials within three years.
Sources
Yadav, S., Lee, R.X., Kajale, S.N., Joy, B.C., Saha, M., Patel, P., Bull, L., Cao, S., Mitragotri, S., Bono, D., & Sarkar, D. (2025). A nonsurgical brain implant enabled through a cell-electronics hybrid for focal neuromodulation. Nature Biotechnology. DOI: 10.1038/s41587-025-02809-3
MIT News (November 5, 2025). New therapeutic brain implants could defy the need for surgery. Massachusetts Institute of Technology.

Jane holds a BSc in Biology from the University of Regina and a Master of Science in Bioscience, Technology and Public Policy from the Univesity of Winnipeg. Her reporting interests include Life Sciences, Physical Sciences and the Cosmos.